
The Cannabis Revolution
About five years ago, the level of research and technology in the fast-growing cannabis market helped drive major advances in product quality and manufacturing methods. Consumers became aware of the various ingestion methods and the importance of high-quality cultivation and refinement methods. At the same time, significant expenditures were invested in researching the medicinal properties of cannabis and in the development of corresponding products.
This market, called cannabinoid-based medicine CBM, is gaining more and more importance compared to the common use of the cannabinoid as a dietary supplement and admixture to common food products and is forcing the increase to the quality level of the available products.
Parallel to this development, a separate market for medical cannabis, essentially flowers with different compositions of the active ingredients, has emerged, whose requirements for the products as a medical active ingredient, is even more clearly defined. There is a new focus on processing the active ingredients contained in the flowers into pharmaceutical end products with higher efficacy and more efficient handling in distribution.
The market development for cannabis products
For the year 2028, the market research institute forecasts an overall European market for cannabis with a volume of 123 billion euros. According to this study, this overall market is divided into a sub-market for medical cannabis (i.e. products with THC content) amounting to 58 billion euros and a further segment for novel food CBD, CBM and “recreational” cannabis (legally permitted smoking marijuana) the respective laws in the respective EU, rest of Europe states, in the USA according to the laws of the respective federal states, in Canada and in other states. Since the legal start of the new agro-pharmacy industry in 2016, products with CBD with a low below 0.2% or no THC content amounting to 65 billion EUR have been produced. In addition, there is the South American market with a forecast volume of 15 billion euros for 2028.
Cannabis Law World Map
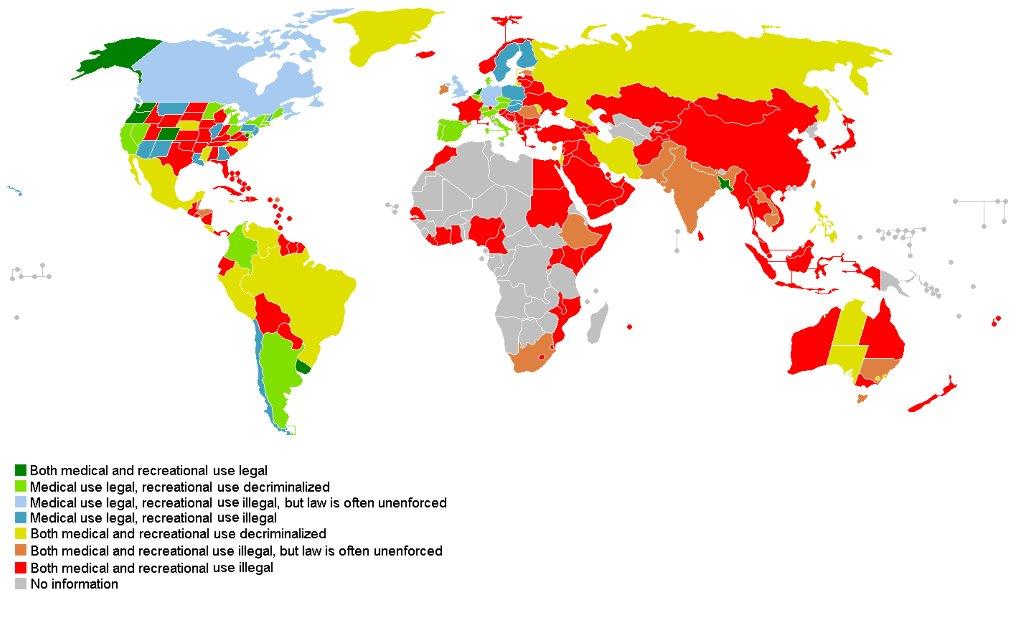
Entwicklungsgesellschaft CPharm International mbH (ECI)
CPharm International brings experience from projects in different countries and has been doing R&D since 2000 . The company knows the current state of relevant research and as well as all the necessary partners to bring all the know-how in the development of complex equipment to international projects.
CPharm International offers assistance in the development of legislation, certification and regulatory requirements, as well as in the construction and operation of production facilities. Recent examples include projects in Afghanistan, Cyprus, Kazakhstan, Kyrgyzstan, Lesotho, Morocco, North Macedonia and Cyprus.
CPharm International also has access to the resources of numerous partner companies that have successfully developed, implemented and marketed products across the spectrum of the cannabis market in the recent past.
ECI Branch Office Republic of Kazakhstan
|
Kazybek bi Ulitsa 1260 KZ-050000 Almaty Tel +7-7756410188 Mr. Zhannur Sultanbekov, Director |

|
Partners of ECI
| Bonglobal Deutschland GmbH |

|
| Prinz Karl Thurn und Taxis Management AG |

|
| Construction of the most modern and largest pharmaceutical production plant |

|